-
Happy Birthday ICMag! Been 20 years since Gypsy Nirvana created the forum! We are celebrating with a 4/20 Giveaway and by launching a new Patreon tier called "420club". You can read more here.
-
Important notice: ICMag's T.O.U. has been updated. Please review it here. For your convenience, it is also available in the main forum menu, under 'Quick Links"!
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
Water Aeration, Simple
- Thread starter B. Friendly
- Start date
Incognegro
Member
Think your waterfall produces more DO in the water than this? http://www.youtube.com/watch?v=eNMTUBtynSY&feature=mfu_in_order&list=UL Doubt it.
WOW! Very impressive!
 Although I do believe 5microns may be a tad small for a DWC using anything other than pure synthetics, but just my opinion; simply because many particles in fertilizers will likely be larger than 5microns, so may not be absorbed properly... (I may be wrong though) I DO like that fog though... almost like a under water aero system..lol!
Although I do believe 5microns may be a tad small for a DWC using anything other than pure synthetics, but just my opinion; simply because many particles in fertilizers will likely be larger than 5microns, so may not be absorbed properly... (I may be wrong though) I DO like that fog though... almost like a under water aero system..lol!
I'm thinking I may end up having to get a couple of those round airstones... I've got 2 different kinds of long stones, and with my newest setup I def need a great airstone.... will figure something out..
exactly. when you fill up a bucket lots of water is exposed to air because of increased water surface during the pour. got little to do with the fall itself. all is needed is calculate surface area...  it isn't about forcing air into water by force but rather about letting water just so slightly have a breath or a touch of air
it isn't about forcing air into water by force but rather about letting water just so slightly have a breath or a touch of air
exactly. when you fill up a bucket lots of water is exposed to air because of increased water surface during the pour.
And the water-fall effect is a pour that never ends. In my system you can see 2 returns and the return from the chiller as well, 3 water falls in one rez. And of course with a water fall design you have to have oversized return lines, so you're talking another 6 feet on each side of my system where there is a lot of area as well.
I doubt there is any practical air-stone set-up that would match it as far as D.O. Somebody linked a video of a compressed air system used in industrial setups that probably cost a few thousands dollars but there's a point where you have to get real.
Hey JF,
Why dont you test the DO in your solution so the non-believers can see the PPM of D'O in your solution. I know you can buy test strips & they are expensive but im not sure how accurate they actually are, but a £5 for one test its unlikely they are totally useless, theve been on the market for a fair few years now. My bet is your over the recommended 30ppms of D'O by quite a bit. its 30ppms-100ppms we are looking for. I never did get an answer about Toxic levels of D'O, or if thats even possible, but sense says it is. G'Luck bro! Im gonna try some, Does anyone have a recommended maker, tried & tested with the test strips? or are they crap?
Im gonna try some, Does anyone have a recommended maker, tried & tested with the test strips? or are they crap?
Why dont you test the DO in your solution so the non-believers can see the PPM of D'O in your solution. I know you can buy test strips & they are expensive but im not sure how accurate they actually are, but a £5 for one test its unlikely they are totally useless, theve been on the market for a fair few years now. My bet is your over the recommended 30ppms of D'O by quite a bit. its 30ppms-100ppms we are looking for. I never did get an answer about Toxic levels of D'O, or if thats even possible, but sense says it is. G'Luck bro!
Hey JF,
Why dont you test the DO in your solution so the non-believers can see the PPM of D'O in your solution. I know you can buy test strips & they are expensive but im not sure how accurate they actually are, but a £5 for one test its unlikely they are totally useless, theve been on the market for a fair few years now. My bet is your over the recommended 30ppms of D'O by quite a bit. its 30ppms-100ppms we are looking for. I never did get an answer about Toxic levels of D'O, or if thats even possible, but sense says it is. G'Luck bro!Im gonna try some, Does anyone have a recommended maker, tried & tested with the test strips? or are they crap?
I don't think 30ppms of DO in water is possible at normal atmospheric pressures much less 100ppms. As to toxic levels of oxygen, I would assume you could get to a toxic level but not sure the water would hold a toxic level of O2.
i believe 7ppms in standard water.(now is that right), i ws just sugesting the amout of D'O that MJ can use bro thats all, 100ppms's being the maximum, also i was wondering if anything over 100ppm's is toxic or not. The rec's ive seen use 100 as a max & they started at 30ppms, why give this advice if its not even possible?
Ive edited this post 7 times now! lol
What if you pumped a little liquid O2 into there? hehe
im back to the books tommoro on this subject, best set myself straight!
(who's the man wearing the beard in your avatar ?)
Ive edited this post 7 times now! lol
What if you pumped a little liquid O2 into there? hehe
im back to the books tommoro on this subject, best set myself straight!
(who's the man wearing the beard in your avatar ?)
Last edited by a moderator:
You are the first person to ask me who my avatar is lol. Ernest Hemingway is the man with the beard. Here is a chart that I found and its sort of what I base my understanding of DO in water on. However, you have to take into account that as the ppm of the solution is increased by other nutrients the level of DO possible decreases. I have never really looked into why this is but I was told by a consultant with a company that works with large scale hydroponic lettuce farmers. He may be wrong but I have no reason to believe so at this point. Oh and the graph, I almost forgot.
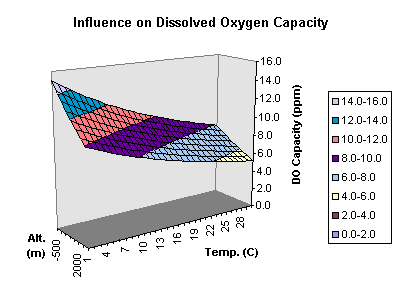
Edit: Oh and cheers on civility.

Edit: Oh and cheers on civility.
That sort of makes sense, it can only hold so much, kind of like a sponge, when you lok at it like that, well you get me. though i stand by what ive read. i just cant bloody remember where, if i find it ill post a link, but the rec D'O level for MJ in Hydro solutions was 30-100 ppms. like i said ill get my head in the books tommoro & get back to you, i have terrible memory at best!
Big Ernie ah, i sort of recognised him but i dont know where the fk from, a book maybe!"
G'Luck & Thanks for the input(Graph)
hey man, were all family here.........Peace!¬)
(nothing on Liquid O2 infusion...Why Not?-try it i mean, people play with Co2, but i suppose it aint quite as dangerous!)
btw that graph confirms my 7ppm! at 68-70f is it? yeah!
Big Ernie ah, i sort of recognised him but i dont know where the fk from, a book maybe!"
G'Luck & Thanks for the input(Graph)
hey man, were all family here.........Peace!¬)
(nothing on Liquid O2 infusion...Why Not?-try it i mean, people play with Co2, but i suppose it aint quite as dangerous!)
btw that graph confirms my 7ppm! at 68-70f is it? yeah!
Hang on, no, that cant be right man. That chart cant be for hydro solutions at all, look where the max DO Vs temps is! na i canna believe that one, whats your take on it here? Thats standard water is it?
Where is that graph from?, 3D, its a mad looking thing!
Anyway add H2o2 & you'd hit those 30-100ppms, why i mentioned the test strips, from the same makers as OXY+
Where is that graph from?, 3D, its a mad looking thing!
Anyway add H2o2 & you'd hit those 30-100ppms, why i mentioned the test strips, from the same makers as OXY+
here ya go man!
#
http://www.indigo.com/test-strips/peroxide-test-strips.html
And i quote- "Very effective for sanitation".
G'Luck Man!
#
http://www.indigo.com/test-strips/peroxide-test-strips.html
And i quote- "Very effective for sanitation".
G'Luck Man!
H2O alone can only hold so much DO, and in that state it is considered at the oxygen saturation point. Increasing the oxygen content past saturation, and into what's called supersaturation happens all the time in natural marine settings, as things like simple photosynthesis can increase DO to the supersaturation point.
H2O2 can also bring water, at any temperature, and at any barometric pressure, into supersaturation.
H2O2 can also bring water, at any temperature, and at any barometric pressure, into supersaturation.
H2O2 can also bring water, at any temperature, and at any barometric pressure, into supersaturation.
Let's say I have 100 liter rez, how much h202 and how often would I need it to bring it to supersaturation?
And the water-fall effect is a pour that never ends. In my system you can see 2 returns and the return from the chiller as well, 3 water falls in one rez. And of course with a water fall design you have to have oversized return lines, so you're talking another 6 feet on each side of my system where there is a lot of area as well.
I doubt there is any practical air-stone set-up that would match it as far as D.O. Somebody linked a video of a compressed air system used in industrial setups that probably cost a few thousands dollars but there's a point where you have to get real.
So what your waterfall never ends? Make bubbler and bubbles don't end too. As said earlier - SURFACE is that matters. Once again, you do not need water to fall and crush. Suppose you pump x gallons of water per hour through a pipe positioned 10inch over the res so water falls down. Now use the same pump, pump the same amount of water but instead of letting it fall let it flow over a large surface into the res (rez wall for example) and measure DO. This is simple mathematics of surface area. Waterfall will be great if it is from high above but it is noisy which is really unnecessary.
And the question about that industrial setup was if it is better for aeration then pipie waterfall and the answer is sure it is. Nothing to do with its price.
I don't think you're getting that the water fall creates more surface. Just like a rolling wheel covers a lot of surface.
Last edited by a moderator:
Yes!!!! I love all this stuff.....
When you bubble oxygen into water, the oxygen molecules basically "kick out" the CO2 molecules in a very complex chemical reaction that I once saw in a pond ecology journal.....this also raises the pH into the alkaline range.....I once took a glass of water and tested the pH.....then I bubbled pure medical grade oxygen into the glass of water and it immediately became very alkaline, I think around pH 8 or 9.....also if your water gets too hot it loses the ability to hold oxygen molecules.....
Those CO2 tubes are laser-drilled with thousands of microscopic pores, so you can use them for your oxygen experiments as well.....
Probably I should say that the use of compressed oxygen tanks can lead to a catastrophic event.....never expose a stream of oxygen to an open flame.....also you should never pass a CO2 tube in front of your nose because you want to know "what CO2 smells like." Trust me, never ever do this. Your brain will suddenly think it is drowning because of the momentary lack of oxygen molecules in your nose.
From everything I have read, you would need an extremely high Dissolved Oxygen level before the roots would become damaged.....just like you would need an extremely high level of CO2 before the stomata begin to close in the leaves.....basically the roots are just leaves that have adapted to Oxygen and not CO2 (a simple way to think about it).....i remember finding a study that did test the ultimate level of CO2, but I can't remember now what that level was, but I probably posted about it somewhere around here.....
I guess you would have to try pure oxygen aeroponics to find out the ultimate level of Oxygen that the roots could withstand.....
And yes I think the falling water effect is better than the bucket of water + airstone, unless you have a really fancy ceramic air-stone
that is designed for industrial purposes.....i have a picture of one of those in my gallery.....
 kind regards from guineapig
kind regards from guineapig 
When you bubble oxygen into water, the oxygen molecules basically "kick out" the CO2 molecules in a very complex chemical reaction that I once saw in a pond ecology journal.....this also raises the pH into the alkaline range.....I once took a glass of water and tested the pH.....then I bubbled pure medical grade oxygen into the glass of water and it immediately became very alkaline, I think around pH 8 or 9.....also if your water gets too hot it loses the ability to hold oxygen molecules.....
Those CO2 tubes are laser-drilled with thousands of microscopic pores, so you can use them for your oxygen experiments as well.....
Probably I should say that the use of compressed oxygen tanks can lead to a catastrophic event.....never expose a stream of oxygen to an open flame.....also you should never pass a CO2 tube in front of your nose because you want to know "what CO2 smells like." Trust me, never ever do this. Your brain will suddenly think it is drowning because of the momentary lack of oxygen molecules in your nose.
From everything I have read, you would need an extremely high Dissolved Oxygen level before the roots would become damaged.....just like you would need an extremely high level of CO2 before the stomata begin to close in the leaves.....basically the roots are just leaves that have adapted to Oxygen and not CO2 (a simple way to think about it).....i remember finding a study that did test the ultimate level of CO2, but I can't remember now what that level was, but I probably posted about it somewhere around here.....
I guess you would have to try pure oxygen aeroponics to find out the ultimate level of Oxygen that the roots could withstand.....
And yes I think the falling water effect is better than the bucket of water + airstone, unless you have a really fancy ceramic air-stone
that is designed for industrial purposes.....i have a picture of one of those in my gallery.....
 kind regards from guineapig
kind regards from guineapig 
Wish the guy with the D.O. meter was still around. He worked in some kind of a lab that had it and he borrowed it. They cost somewhere around $400 I think, I've been tempted in the past to buy but it's just above the level of stupid I'm willing to go to prove a point on a pot board.
Another link to DO levels in water at various atmospheric pressures ~> http://www.google.com/url?sa=t&sour...ocSZAw&usg=AFQjCNE6kDRzqhElLrMj1cxFUyj5gyvyWQ
Baba you have any info on supersaturation of O2 in nutrient solutions?
Baba you have any info on supersaturation of O2 in nutrient solutions?
... I always thought, well i know that Co2 raises PH, never knew about pure O2 raising ph, intersting & something i know nothing about! where did you get that info Guiniepig? ill have to look into it! I wonder the DO level under Niagra or Vic Falls or somewhere, anyone? The Ultimate Waterfall! I dont doubt this action is better than pumping it in when i think of large waterfalls! its like white bubbling clouds! Its just too bloody noisey! Stealth is out the window!
I always thought, well i know that Co2 raises PH, never knew about pure O2 raising ph, intersting & something i know nothing about! where did you get that info Guiniepig? ill have to look into it! I wonder the DO level under Niagra or Vic Falls or somewhere, anyone? The Ultimate Waterfall! I dont doubt this action is better than pumping it in when i think of large waterfalls! its like white bubbling clouds! Its just too bloody noisey! Stealth is out the window!
I dont get that chart sorry Zee, does it really apply to us?. its hydro solution that we need. it only goes upto 14c anyway, N/a.
I dont get that chart sorry Zee, does it really apply to us?. its hydro solution that we need. it only goes upto 14c anyway, N/a.
Last edited:
Im getting away with 10lpm with 4 standard cheapo airstones & a 70 litre res. plants love the shit & uptake seems great! If i put in H2O2 i get One Hell of a Boost, you can see nute uptake is trebled! again the use of H2o2 puts me in that 30-100ppm range!(im gonna get some test kits/strips!)
Latest posts
-
-
-
-
-
Australian Dead Head (mullumbimby madness x Neville's haze)?
- Latest: StickyBandit
Latest posts
-
-
-
-
-
Australian Dead Head (mullumbimby madness x Neville's haze)?
- Latest: StickyBandit


